
The Council for Medical Schemes recently published an insightful study in the American Journal of Biomedical Science and Research titled ‘Profiling Dependants Joining Medical Schemes: An Analysis of Factors Influencing Enrolment Decisions.’
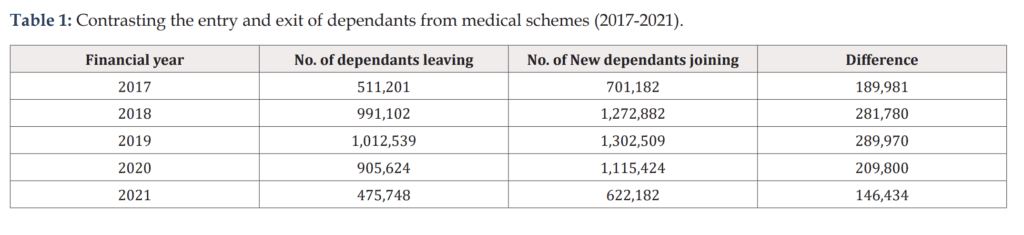
The study provides a fresh perspective in the ongoing debate between ‘medical aids’ and ‘cheap medical aid.’ From 2017 to 2021, an average of 1.1 million new dependants joined medical schemes each year, with newborns making up 23% and children under five accounting for 35%.
The authors suggest that by understanding enrolment decisions, medical schemes can tailor their strategies to the specific needs of dependants, potentially leading to higher enrolment rates and improved healthcare outcomes.
Enrolment in a medical scheme is a complex process influenced by multiple factors, including affordability, age, and healthcare needs.
Interestingly, in 2019, medical schemes collectively saw a high number of new dependants join the scheme at 1.3 million. However, in 2021, this number dropped to 622,000, likely due to the economic impact of COVID-19.
Closed schemes tend to enrol more new dependants below the age of 5, while open schemes typically enrol more beneficiaries aged 60 years.
The study also revealed that costs, availability of cost-sharing mechanisms, and financial incentives influence dependants’ decision to enrol in medical schemes.
To expand health insurance coverage, interventions should prioritize the healthcare needs of children and the elderly.
Recent Posts

SAHPRA SAPC Regulatory Enforcement: Addressing Unregistered GLP-1 and GIP Products

Substandard Medicines in South Africa: Regulatory Challenges and Economic Implications
