
The Role of Transparency in Coverage Data
In the realm of healthcare, the importance of transparency cannot be overstated. A recent study by Benjamin L. Chartock, PhD; Kosali Simon, PhD; Christopher M. Whaley, PhD, has shed light on how transparency in coverage (TiC) data can significantly influence prices for common healthcare services. The researchers examined TiC price data from Humana, a major national insurer in the USA, highlighting the potential uses of such data for future research.
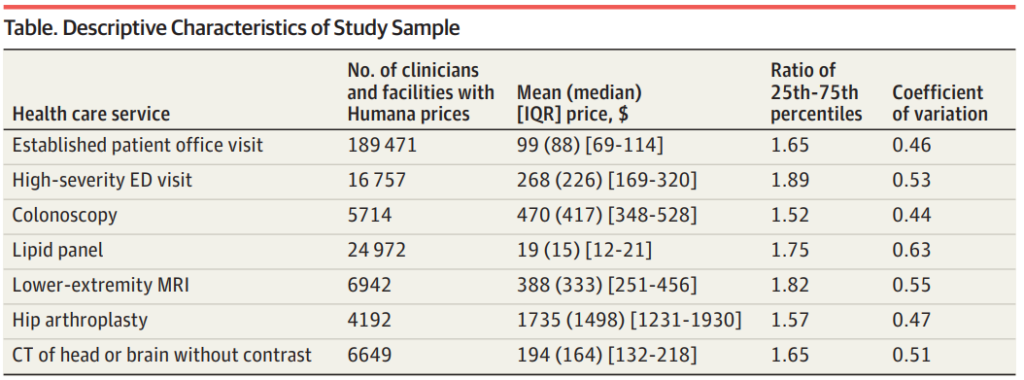
Unravelling Price Variation
The study’s findings revealed substantial price variation for common healthcare services. The researchers focused on seven procedures, including both ‘shoppable’ and ‘non-shoppable’ services. Interestingly, the variation in prices for these services was similar, regardless of their ‘shoppability’. The study also noted that mean county-level prices were lowest in the central US and Florida, with higher prices observed in the upper-Midwest and Southeast.
The findings of the study make a compelling case for the necessity of further research into the factors that drive price variation in healthcare. The topic of whether or not these prices accurately reflect worth, or whether or not they are an indication of inequalities in market power and negotiation leverage, will need to be investigated more in the future. If the second possibility is correct, then there is an even greater requirement for regulations that guarantee competitive healthcare markets.
In conclusion, the study highlights the potential of informed healthcare consumerism as a lever for managing costs and enhancing patient happiness. It also opens up new possibilities for the use of TiC data in larger settings, which opens up new avenues for using TiC data.
Recent Posts

Prioritizing a Comprehensive Medtech Market Access Strategy Through Stakeholder Engagement

Potential for Advancements in HIV Weekly Treatment Options

