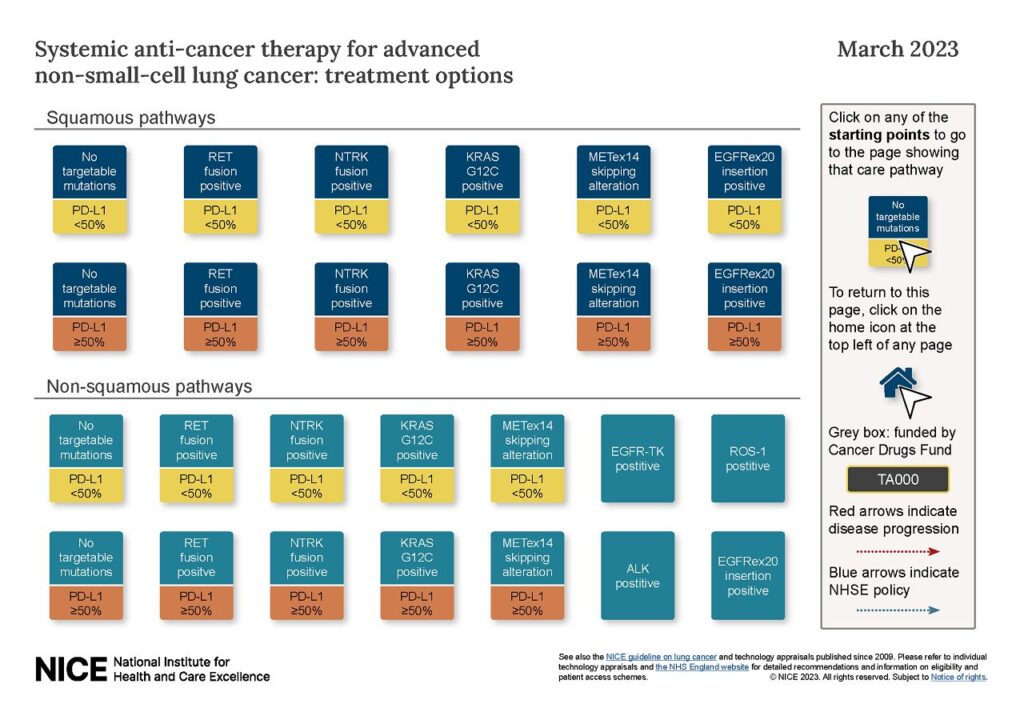
NICE guidelines have added the guidance on mobocertinib to the systemic anti-cancer therapy treatment pathways for advanced non-small-cell lung cancer.
They would like your feedback on how the treatment pathways present content from different sources (NICE technology appraisal guidance, guideline recommendations, and expert input). To comment, go to the link below on systemic anti-cancer therapy for advanced non-small-cell lung cancer and fill in the pop-up survey.
Recent Posts

Advancing Hemophilia Treatment through FDA Approval of Innovative Prophylaxis
FDA Approval Hemophilia Treatment expands meaningful choices for people with hemophilia A and hemophilia B whose needs have long exceeded available therapies. Pfizer’s HYMPAVZI (marstacimab-hncq) now offers a once-weekly subcutaneous non-factor option to adults and adolescents 12 years and older ...

Potential for Advancements in HIV Weekly Treatment Options
HIV Weekly Treatment Options took a major step forward as Gilead and Merck reported that their investigational once-weekly combination of islatravir and lenacapavir successfully met the primary endpoint in two Phase 3 trials. The oral regimen maintained viral suppression in adults already control...

Tovorafenib Clinical Assessment Outcomes and Methodological Challenges
The Tovorafenib clinical assessment conducted under the EU Joint Clinical Assessment process has exposed critical shortcomings in the evidence base for treating BRAF-altered pediatric low-grade glioma. Despite the therapy’s potential, reviewers concluded that robust comparative data is absent for...