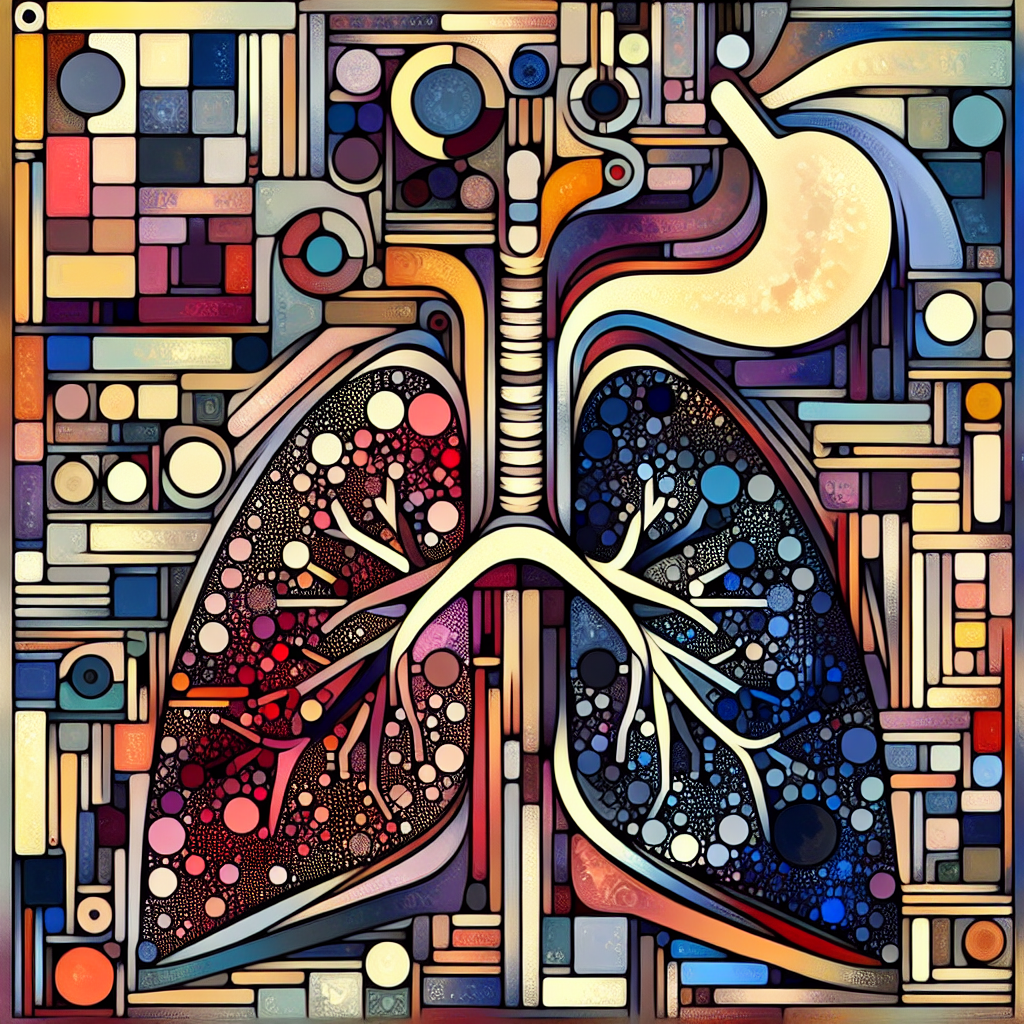
💡 Are you prepared for the surge in elderly care costs projected for 2025?
A recent report from Zorginstituut Nederland reveals that the financial burden on the Dutch health system is set to rise significantly due to demographic shifts and increased demand for complex care. As policymakers navigate these challenges, the insights address crucial implications for health economics, reimbursement strategies, and sustainable access to care.
Don’t miss out on understanding how these trends could shape the future of healthcare. Dive into the full article for comprehensive insights!
#SyenzaNews #HealthEconomics #HealthcarePolicy #DigitalTransformation