
The Rising Challenge of Hypertension
Hypertension management, a significant risk factor for cardiovascular and kidney diseases, is a leading cause of preventable death and disability globally. With more than 100 million people affected in the US alone, the healthcare costs associated with hypertension surpassed $130 billion between 2003 and 2014. Despite the availability of affordable medications and lifestyle interventions, blood pressure control rates continue to decline.
Pharmacist Interventions: A New Approach to Hypertension Management
Recent research suggests an innovative solution: pharmacist-led interventions. Pharmacists, who often see patients more frequently than physicians, are well-positioned to screen and manage hypertension. Numerous clinical trials have demonstrated improved hypertension outcomes through pharmacist-led case-finding and prescribing interventions.
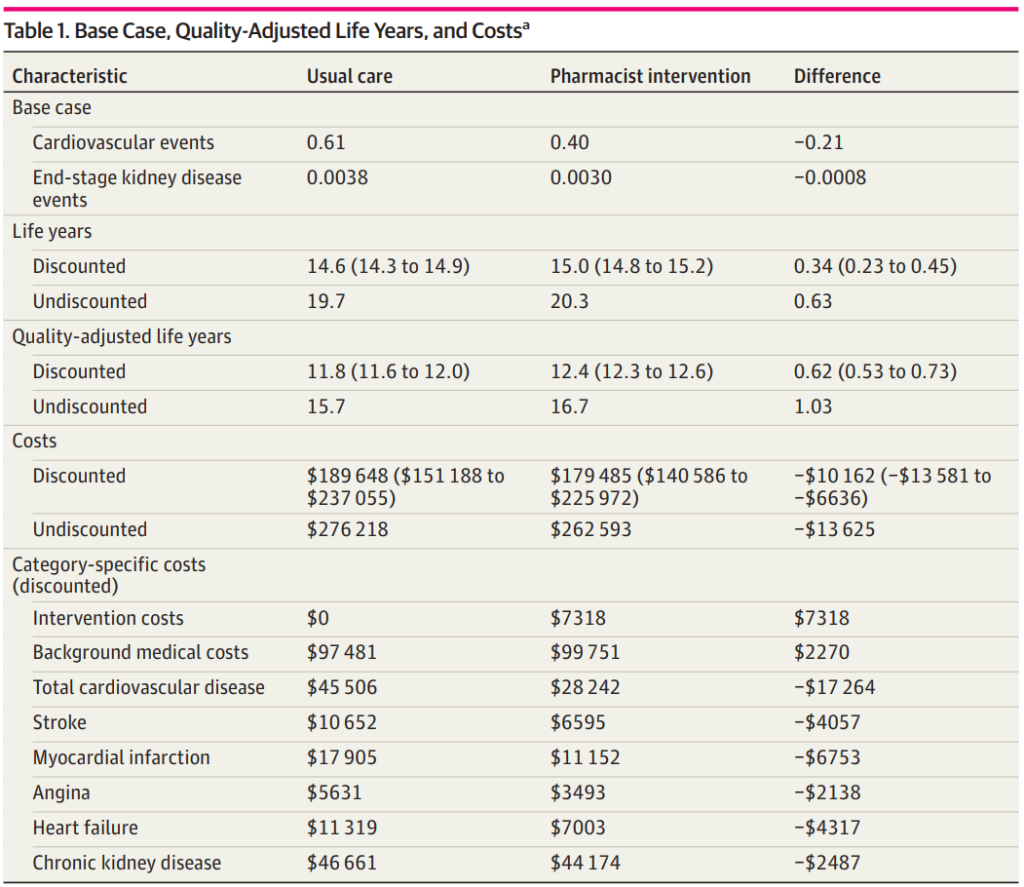
A cost-effectiveness analysis of implementing pharmacist prescribing for hypertension management in the US has yielded promising results. The pharmacist intervention was associated with 2100 fewer cases of cardiovascular disease and 8 fewer cases of kidney disease per 10,000 patients. Furthermore, it resulted in overall cost savings of $10,162 per person.
