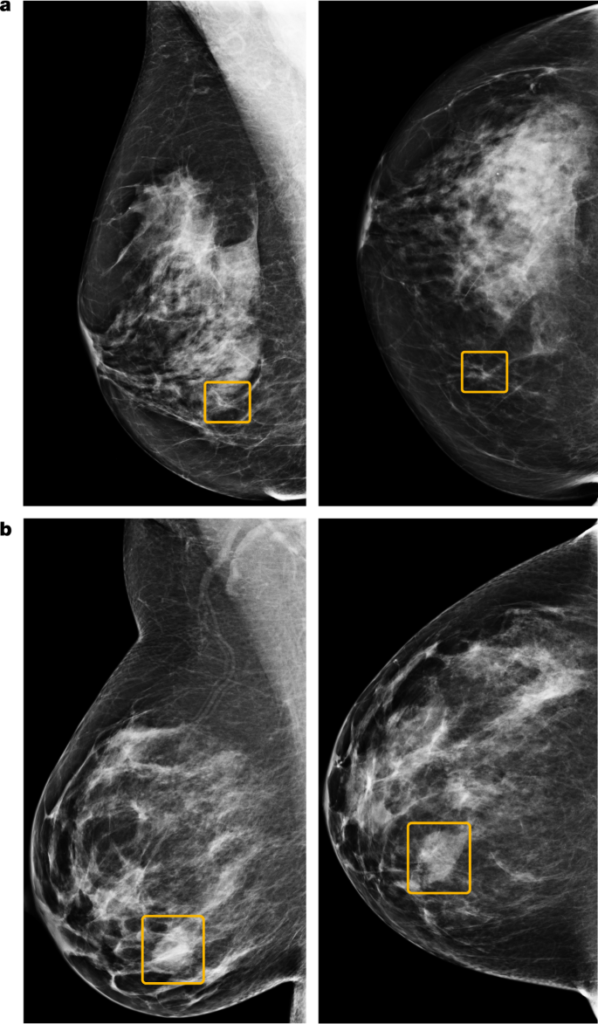
Breast cancer is the second leading cause of cancer death in women. Early identification can ease this to ensure early access to treatment. Developed nations have vast mammography screening programs, but interpreting the images is still hard. The accuracy of cancer detection varies widely among experts, and the potential for improvement is significant. False positives can lead to patient anxiety, unnecessary follow-up, and invasive diagnostic procedures. Sometimes screening cannot identify cancers until they are more advanced and less amenable to treatment.
Artificial Intelligence (AI) can overcome this challenge. Studies show it can outperform human experts in many medical image analysis tasks. This leads us to evaluate AI’s potential to augment the early identification process of breast cancer through high-quality images.
Researchers recently tested a new AI system that predicts breast cancer using large datasets from the UK and the USA. The system outperformed radiologists in daily predictions, according to the researchers. They confirmed these observations with an independently conducted reader study. The system could generalize across different screening populations and settings without additional training data.
The AI system might reduce the workload involved in the double-reading process used in the UK while preserving the standard of care. The system could also provide automated, immediate feedback in the screening setting. This technology can sort cases at the risk level, making it possible to screen efficiently even with a limited workforce.
Overall, the AI was better than radiologists at identifying breast cancer. These results held across two large datasets that represent different screening populations and practices. However, the optimal use of the AI system within clinical workflows remains to be determined.
Cover image taken from the journal article.