
Introduction
Global eye health is a crucial aspect of overall well-being and quality of life, with significant implications for disease reduction, productivity, and education access. While progress has been made through global initiatives, challenges persist. However, the rise of digital health and artificial intelligence (AI) presents innovative opportunities in eye care, particularly in increasing accessibility and aiding clinical decisions with data. To overcome implementation barriers, the establishment of a global digital eye health task force is proposed, which would coordinate funding, infrastructure development, and ensure equitable access to these technologies, ultimately advancing global eye health. AI enhances global eye health access and informs positive change in clinical decision making.
The Power of AI enhances Global Eye Health Access
Digital health, the convergence of medicine and technology, has rapidly evolved in recent years. The World Health Organization (WHO) recommends evaluating digital health tools based on their benefits, harms, feasibility, and equity. Key innovations in this field include AI, telemedicine, virtual reality, and wireless technologies, which have integrated into clinical practices for diagnosis, management, and prognosis. AI, particularly deep learning, has shown remarkable effectiveness in analyzing complex data such as images and videos. In ophthalmology, AI-driven diagnostic tools have proven successful in diagnosing conditions like diabetic retinopathy and glaucoma through medical imaging, gaining recognition and approval from regulatory bodies like the FDA.
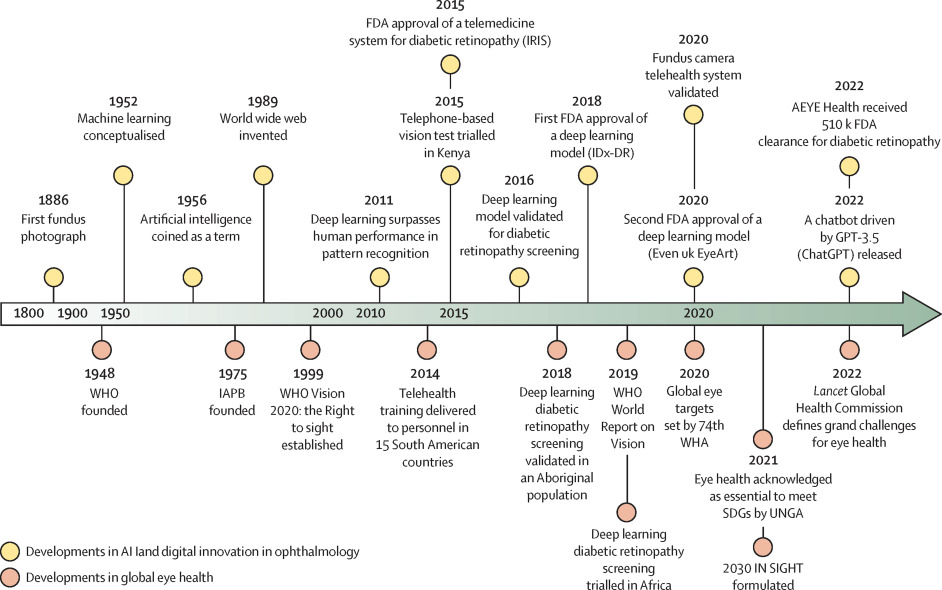
Figure 1: Timeline of the progress of global eye health initiatives compared with the development of digital health and artificial intelligence applications in eye health.
The Need for a Global Digital Eye Health Task Force
To ensure the seamless integration of digital health tools into eye care systems worldwide, the establishment of a global digital eye health task force is proposed. This task force, comprising representatives from every world region, would focus on setting standards, providing training, and evaluating new technologies for eye care. Inspired by successful initiatives like the American Academy of Ophthalmology’s Telemedicine Task Force and the International Agency for the Prevention of Blindness (IAPB) Technology Taskforce, this global task force aims to guide nations in assessing the suitability of technology, ensuring safety, and integrating it into existing health systems. By adopting a collaborative approach, nations can set realistic goals based on their unique economic and healthcare landscapes, while also learning from each other’s experiences. This approach would particularly benefit low- and middle-income countries (LMICs) by providing access to cutting-edge technologies and expertise.
Conclusion
The integration of digital health and AI into global eye health has the potential to transfigure the field, enhancing accessibility and aiding clinical decision-making. By establishing a global digital eye health task force.
Recent Posts

CHMP Recommendations 2026: New Approvals and Implications for Health Economics

Clinical Trial Monitoring: Insights from Q1 2026 EU/EEA Report

