Introduction to the ACTFAST-3 Study
The Anesthesiology Control Tower (ACT), a real-time telemedicine decision support system, was tested in the ACTFAST-3 Randomised Clinical Trial. The study aimed to evaluate the feasibility of the ACT and its impact on two quality of care measures: postoperative hypothermia and hyperglycemia.
The ACTFAST-3 study, a single-centre pilot superiority RCT, took place in the USA from April 2017 to June 2019. The study involved all patients aged 18 years and above who had undergone surgery. However, the study did not include patients if the ACT staff did not staff more than 50% of their case duration or if they did not suffer from diabetes (for glycemic outcomes).
Results and Implications
The pilot study, involving 26,254 patients, found that the telemedicine decision support intervention did not significantly alter the two quality of care outcomes. While there was a slight increase in intraoperative glucose measurement in the intervention group, there was no difference in the frequency of treatment for hyperglycemia. This lack of difference could explain the unchanged postoperative hyperglycemia rates.
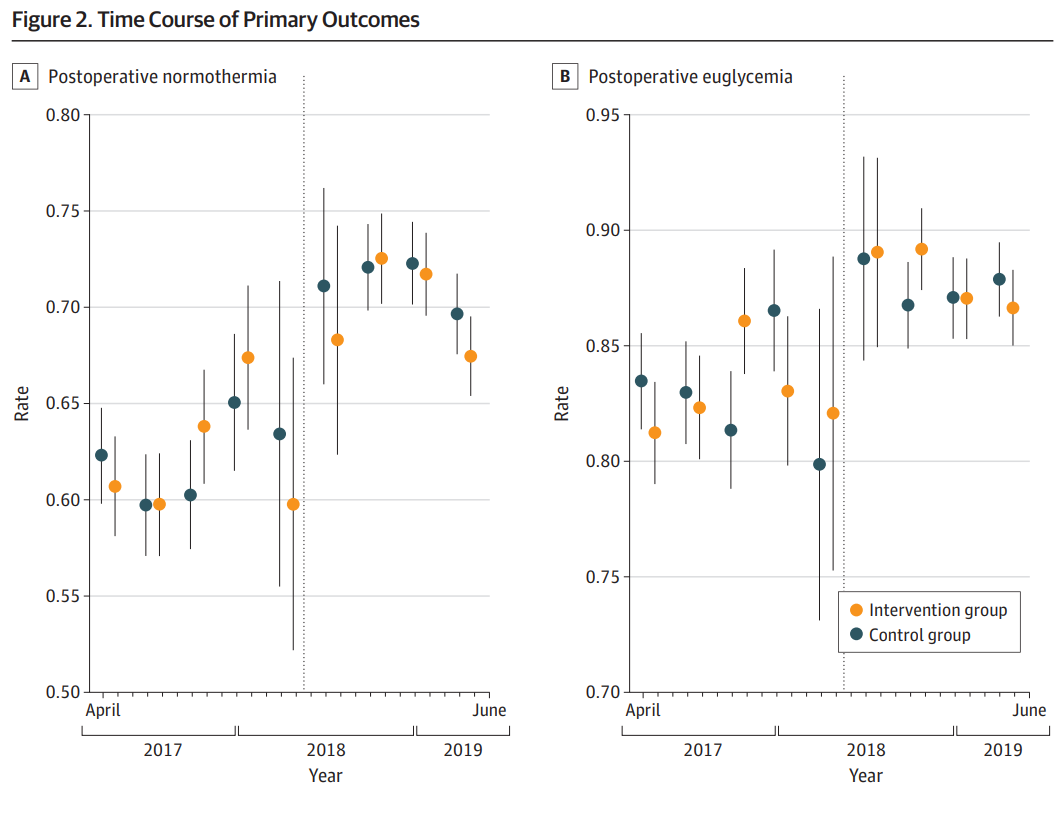
The study also highlighted potential reasons for these results. For instance, the high adherence to each measure at baseline made significant improvements challenging. Also, the trial noted the impact of the Hawthorne effect, where changes in behaviour occur due to the awareness of being observed.
The study concluded that further optimization of clinical decision support and workflows might help the intraoperative telemedicine program achieve improvement in targeted clinical measures.
Conclusion:
The ACTFAST-3 study offers crucial insights into the application of telemedicine in anesthesiology, yet it underscores the complexity of enhancing care quality through such interventions. Despite the implementation of the Anesthesiology Control Tower, improvements in postoperative hypothermia and hyperglycemia were not evident. This outcome suggests that high baseline adherence to quality measures and behavioural modifications due to the Hawthorne effect may dilute the impact of telemedicine interventions. Consequently, to realise the potential benefits of real-time decision support systems, we must refine clinical support tools and streamline operative workflows. The findings prompt a re-evaluation of strategies within the telemedicine framework to ensure that technological advancements translate into tangible improvements in patient care.
Recent Posts

Expanded GLP-1 Medication Access Through Formulary Updates

European Pharmacists Preparation for Health Crisis Response

